

As 2023 draws to a close, Tsingke remains dedicated to its mission: “Biotech for a Better World.” Driven by technological innovation, we have continued to advance the TSINGKE GENE FACTORY. Let’s revisit the milestone moments that defined our year!
As a pioneer of the "Gene Factory" concept, Tsingke is committed to technology-led growth. We have achieved significant breakthroughs in all stages of factory development. By enabling fully automated and intelligent gene synthesis, we continue to empower scientific research and expand the reach of biotechnology applications.
In April 2023, Tsingke hosted the Nucleic Acid Drug Development Forum, marking the official launch of our Jintan Synthetic Biology Industrial Base. During the event, we unveiled the TsiKer™ high-loading synthesizer. This breakthrough is a vital catalyst for the TSINGKE GENE FACTORY, driving high-quality progress in the nucleic acid drug industry.


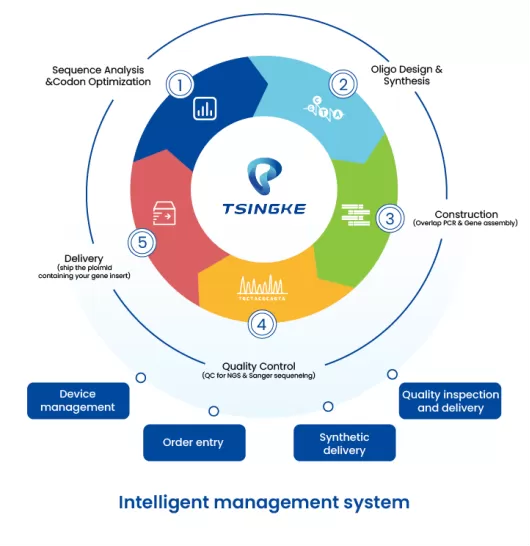
In August 2023, we completed the digital deployment of our "Gene Factory" and launched a fully online, intelligent management system. Our gene synthesis capabilities now ensure stable and rapid turnaround, with short fragment synthesis delivered in as little as 72 hours.
In September 2023, we launched our high-throughput nucleic acid extractor. This milestone innovation significantly expands the automated production capacity of the TSINGKE GENE FACTORY.

Our Fuzhou Branch sequencing laboratory is now fully operational, while the Guangzhou Branch has officially launched its fast NGS sequencing lab.
Driven by Tsingke’s fully integrated intelligent management system, we continue to provide reliable and rapid gene synthesis. Customers can now expect short-fragment synthesis delivery in as little as 72 hours.
Participated in 16 exhibitions to provide customers with more efficient and intelligent solutions



Received the research of industry, academia and research from various parties and continue to explore and innovate



Completed 95 live online classes to empower customers with knowledgeHelped clients publish 3768 articles in Cell Nature Science and other journals*(*1 January 2023-31 December 2023)
Tsingke remains at the forefront of core technology research and innovation. We are the first biopharmaceutical company in China to successfully industrialize long-fragment gene synthesis. Our leadership in resource integration and technological advancement has earned us widespread recognition and established Tsingke as a trusted industry leader.

On March 23, 2023, Tsingke Biotechnology Co., Ltd. was officially renamed Beijing Tsingke Biotech Co., Ltd.
Looking ahead to 2024, Tsingke remains committed to partnering with you as we move confidently into the future. Guided by our "Tsingke Gene Factory" vision, we aim to pioneer advancements in biosynthesis and drive industry-wide, high-quality development through continuous innovation.